Designed for flexibility, our innovative duct occluder options conform to a variety of duct sizes while achieving complete patent ductus arteriosus (PDA) closure from a pulmonary or aortic approach.
SWITCH TO THE REFERRAL VIEW For more information specific to neonatology regarding PDA closure
AMPLATZER PICCOLO™ OCCLUDER:
FOR PDA CLOSURE IN THE SMALLEST PATIENTS
The Amplatzer Piccolo™ Occluder, which delivers proven PDA closure for patients 700g and up, provides the strength to occlude small ducts, while minimising protrusion into surrounding pulmonary artery and descending aorta.4
Features include:
- Extremely low-profile delivery—4 Fr catheter facilitates delivery in small vasculatures
- Low-profile retention discs and end screw are designed to minimise protrusion into the aorta and pulmonary artery
- Self-expanding device designed for closure of a PDA with length longer than 3mm and diameter smaller than 4mm
- Symmetrical design for either pulmonary or aortic approach
- Tightly woven, single-layer mesh design to minimise residual shunt after placement
- Flexible delivery cable optimises deployment and allows for representative preview of device orientation prior to release
- Predictable placement—disc size and shape designed for predictable positioning in the duct
- Ability to be easily recaptured and redeployed for optimal placement7

INDUSTRY-LEADING AMPLATZER™ PDA OCCLUDERS1
Amplatzer Piccolo™ is the only approved device specifically designed and indicated for PDA closure in premature infants. Only Piccolo™:

PICCOLO™
Has been developed for babies as small as 700gr and ≥ 3 days old

CLINICAL DATA
Has the most clinical data, proving excellent outcomes

SIZE
Offers the widest range of sizes, enabling treatment of most duct sizes

COMPATABILITY
Is compatible with infant size, offering a low profile device and delivery system
LEARN MORE:
Amplatzer Piccolo™ Delivery System (PDS)
A purpose-built delivery system for premature patients receiving the Amplatzer Piccolo™ Occluder to close their patent ductus arteriosus9,10,11
- Used to deliver the most-studied Preemie PDA occluder on the market today, Amplatzer Piccolo™15
- Optimized 45 cm length for the preemie patient in the cath lab or beside in the NCU
- Distal end configured for precise placement of the occluder with an F type ductus
- Delivers consistent and uncompromised safety
- Enables optimal device placement prior to final release of the device
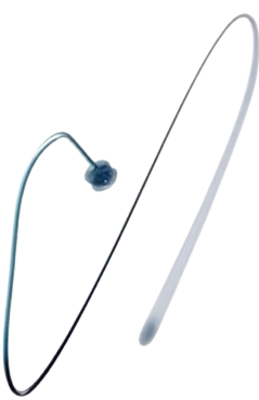
HOW IT WORKS:
Implanting Procedure
The Amplatzer Piccolo™ Occluder implanting procedure is a minimally invasive transcatheter procedure. A small incision is made in the groin for the delivery catheter to advance through the ductus. Once the proper position is located, the occluder is deployed firmly into the ductus. The occluder’s size and shape is specifically designed to fit in the ductus of a premature infant to provide safe and effective PDA closure.
PDS and TVLPC Procedural Comparison
PDA is designed to reduce workflow steps and the accessories required by removing the need for a cathater exchange. PDS has an optimal shape and structure to provide access to the PDA.
PDS PROCEDURAL STEPS
- With a PDF catheter and a floppy atraumatic tip 0.035" Wholey wire, navigate through the right heart and cross the PDA*
- Remove Wholey wire and advance Piccolo Occluder
TVPLC PROCEDURAL STEPS
- With a 4F angled glide catheter and a 0.035-inch floppy atraumatic tip guidewire, navigate through the right heart and cross PDA
- Leave Wholey wire in place across PDA and remove angled glide catheter
- Advance TVPLC over Wholey wire across PDA
- Remove Wholey wire and advance Piccolo Occluder
TV
- Data on file at Abbott.
- Philip R, Waller BR III, Agrawal V et al. Morphologic characterization of the patent ductus arteriosus in the premature infant and the choice of transcatheter occlusion device. Catheter Cardiovasc Interv. 2016;87(2): 310–317. doi.org/10.1002/ccd.26287.
- Schneider DJ, Moore JW. Patent ductus arteriosus. Circulation. 2006;114(17):1873-1882. doi.org/10.1161/CIRCULATIONAHA.105.592063.
- Amplatzer Piccolo™ Occluder Instructions for Use.
- Stout KK, Daniels CJ, Aboulhosn JA et al. 2018 AHA/ACC Guideline for the Management of Adults with Congenital Heart Disease: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation. 2019;139(14):e698–e800. doi.org/10.1161/CIR.0000000000000603.
- Baruteau A-E, Hascoët S, Baruteau J et al. Transcatheter closure of patent ductus arteriosus: past, present and future. Arch Cardiovasc Dis. 2014;107(2):122–132.
doi.org/10.1016/j.acvd.2014.01.008. - Data on File, 302350-001_G Amplatzer™ Duct Occluder CER.
- Saliba Z, El-Rassi I, Abi-Warde MT et al. The Amplatzer Duct Occluder II: a new device for percutaneous ductus arteriosus closure. J Interven Cardiol. 2009;22(6):496–502.
doi.org/10.1111/j.1540-8183.2009.00504.x. - Gruenstein DH, et al. Transcatheter closure of patent ductus arteriosus using the Amplatzer™ Duct Occluder II (ADO II). Catheter Cardiovasc Interv. 2017;89(6):1118-1128. doi.org/10.1002/ccd.26968.
- Windchill 91102617 – Piccolo Delivery System Design Verification Report
- Windchill 91060911 – Design description document (DDD)
- Windchill 91064365 – HFE summary report
- ARTEN600347844_PDS IFU US
- Patent - US 2023/0405269 A1 SINGLE-CATHETER ENABLED PDA ACCESS AND OCCLUDER DEPLOYMENT AND RECAPTURE
- 91061375 - CER